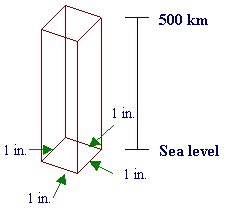
Earth Science: Geology, the Environment, and the UniverseChapter 11: AtmosphereProblem of the WeekA Weighty Matter Every second of every minute of every hour of every day our bodies are being pressed in at all points by the air that surrounds us, and we don’t even notice. We live in an ocean of gases—nitrogen, oxygen, carbon dioxide, water vapor, argon, and traces of other gases in extremely small amounts. (See the pie graph on page 272 of text for proportions.) Because the atoms that make up each of the atmospheric gases has mass, the gravity of Earth attracts the mass of the atoms. This gravitational pull of the air toward Earth causes air pressure. To form a mental picture of "air pressure," picture this—a column of air that is as tall as the atmosphere (500 km at the upper reaches of thermosphere—diagram on page 273) and 1 inch by 1 inch wide (see diagram below). Let’s think about how this affects us. We will use inches for this problem instead of centimeters because one of the standard units for pressure is psi (pounds per square inch).
On Earth, the average pressure at sea level caused by a column of air
such as this is 14.7 pounds per square inch (14.7 psi). This means that
the pressure on that 1-inch by 1-inch square on Earth’s surface is equal
to a weight of 14.7 pounds or 6.62 kilograms. Problem: Since we live at the bottom of this "ocean" of air, each inch
of our bodies has 14.7 psi being exerted upon it. If the average surface
area of the skin of an adult is about 3100 in2, how many total
pounds of pressure are being exerted on his or her body? Why don’t we
implode? How would this amount change as we went up in altitude? Why? |  |